Abstract
- Issue: Novel medicines hold promise for many patients, but rising costs pose growing affordability and equity challenges for health systems worldwide. In the United States, prices are typically higher than in peer countries, and increasing spending strains payers and imposes a financial burden on patients.
- Goal: To identify international pricing and financing strategies with potential relevance for U.S. policymakers.
- Methods: Literature review; case studies of Australia, Canada, England, France, Germany, and Sweden; interviews with policymakers, payer organizations, health system leaders, and researchers.
- Key Findings and Conclusions: Cross-cutting lessons include: 1) systematic use of health technology assessment (HTA) provides a basis for evaluating clinical benefit and informing coverage and pricing decisions; 2) centralized or collaborative negotiations improve bargaining power and support more predictable pricing, even in decentralized health systems; 3) structured pathways that allow early patient access while generating additional evidence, such as managed entry agreements, help control budget impact and financial uncertainty; 4) investments in data and transparency enable more sophisticated financing models; and 5) equity and patient protection are core principles, with each country aiming to minimize out-of-pocket spending. For the U.S., these lessons suggest actionable policy options: strengthening shared evidence assessment, enabling state–federal or multipayer partnerships, continued investment in evidence infrastructure, and embedding safeguards to ensure access for underserved patients.
Introduction
The rising cost of prescription medicines presents a significant challenge to health systems worldwide. High prices strain the budgets of public programs and insurers, limit patient access to needed therapies, and deepen inequities, with lower-income and underserved populations bearing a disproportionate burden. Specialty therapies pose a particular challenge: treatments for oncology, rare diseases, and gene and cell therapies account for only 1 percent to 2 percent of prescriptions but over a quarter of drug spending in many high-income countries. Many of these therapies offer significant clinical advances. Yet, in the United States, where prices are typically higher than in peer countries, rapid spending growth is increasing pressure on public payers and employer-sponsored insurance. This is forcing difficult trade-offs in coverage and creating financial obstacles for patients.
Recognizing these concerns, U.S. policymakers and payers have begun experimenting with innovative pricing and financing mechanisms — outcome-based contracts, subscription pricing, and reinsurance pools — but questions remain about their scalability and long-term sustainability within the fragmented U.S. health system.
Other high-income countries have developed diverse approaches to managing pharmaceutical costs within more centralized health systems with varying success. Drawing on the literature and stakeholder insights, this issue brief documents innovative pricing and financing models used by selected countries — Australia, Canada, England, France, Germany, and Sweden — to manage access and ensure affordability of high-cost medicines. The table below summarizes selected approaches across these countries. Examining their outcomes, feasibility, and equity implications highlights which strategies may offer actionable insights for the U.S.
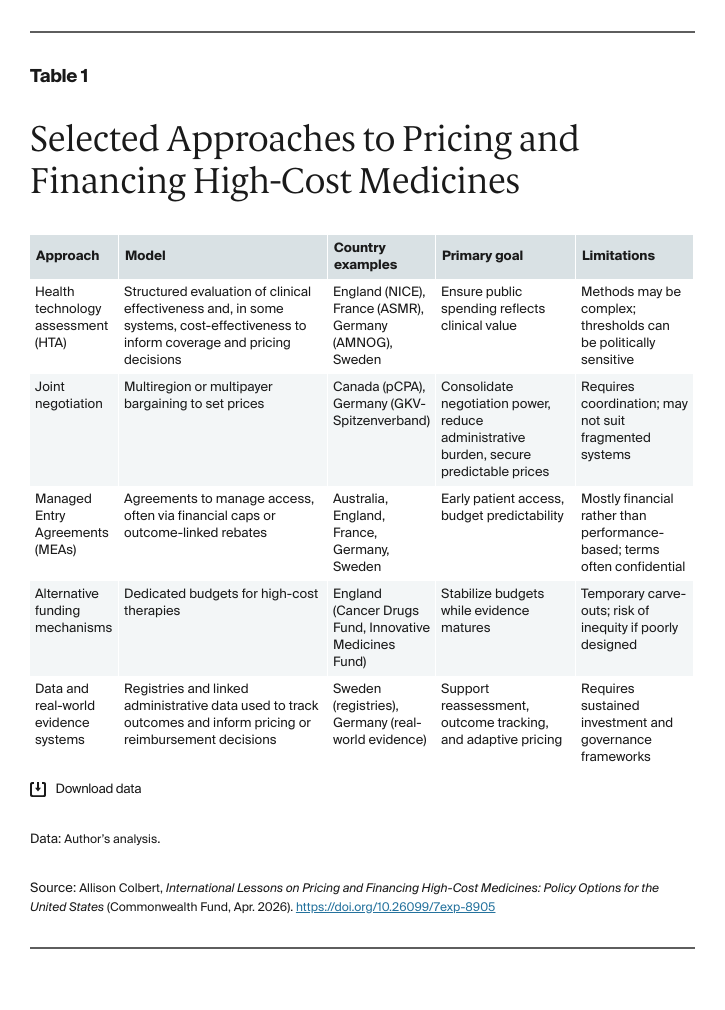
Key Findings
International experience shows that achieving affordability and access for novel, high-cost medicines requires deliberate, system-level policy design. Across Australia, Canada, England, France, Germany, and Sweden — countries that all provide universal health coverage but vary in how they deliver care — policymakers have developed policy frameworks to negotiate effectively with manufacturers, protect patients from catastrophic costs, and enable adoption of transformative therapies while managing budgetary risk. These systems illustrate different policy approaches designed to balance access, affordability, and budgetary sustainability. Despite differences in national context and institutional structure, several cross-cutting processes are relevant to the United States. (Appendix 1 provides a snapshot of country approaches, while Appendix 2 summarizes lessons for the U.S.).
Health technology assessment frameworks and evidence-based thresholds build transparency and trust.
Clear health technology assessment (HTA) frameworks help determine which medicines offer sufficient clinical value to merit public investment and provide a structured basis for pricing and financing decisions. All six countries use formal HTA processes to assess clinical effectiveness, comparative benefit, and, in some cases, cost-effectiveness of new medicines, though HTA’s role in final coverage and pricing varies.
Notable examples include France’s Amélioration du Service Médical Rendu (ASMR) ratings, which classify the degree of added therapeutic benefit relative to existing treatments, and England’s cost-effectiveness thresholds applied by the National Institute for Health and Care Excellence (NICE). Germany’s AMNOG process similarly anchors price negotiations in a formal assessment of added clinical benefit based on structured clinical evidence. Together, these frameworks create more consistent and predictable decision-making, even where final prices remain confidential.
At the supranational level, the European Union’s new HTA Regulation, phasing in from 2025, aims to harmonize clinical assessments across Member States while leaving pricing and reimbursement decisions to national authorities. This reflects a broader trend toward shared evidence generation to reduce duplication and speed access while preserving local policy discretion.
Interviewees emphasized that clear HTA rules reduce politicization and build trust. However, many systems deliberately separate HTA from pricing negotiations: while clinical value assessments are public, final prices often rely on confidential discounts or rebates. This separation preserves HTA neutrality but creates a tension between public accountability and commercial nondisclosure.
For the U.S., which lacks a national HTA body, more systematic evidence appraisal — potentially through independent organizations such as the Institute for Clinical and Economic Review (ICER), regional consortia, or payer collaboratives — could strengthen legitimacy and consistency in coverage decisions. While the U.S. is unlikely to adopt a central HTA authority, expanding credible, shared evidence standards could help balance transparency with negotiation flexibility and support more coherent approaches to high-cost medicines.
Collaborative negotiations are feasible even in decentralized multipayer health care systems.
All six countries consolidate bargaining power through national or joint negotiations. France and Germany conduct price negotiations for high-cost medicines at the national level, with Germany’s statutory health insurance funds (covering most of the population) negotiating centrally through the GKV-Spitzenverband. NHS England sets coverage terms and negotiates prices, providing a centralized framework within a devolved system.
In federal systems with shared responsibilities across levels of government, coordination mechanisms play a similar role. In Canada, the pan-Canadian Pharmaceutical Alliance (pCPA) negotiates on behalf of provinces and territories. Australia’s Pharmaceutical Benefits Scheme (PBS) secures consistent pricing for community medicines, with states responsible for hospital procurement, and the Highly Specialised Drugs Program subsidizes high-cost medicines requiring specialist management. Sweden also has a regionally structured system in which collaborative negotiation mechanisms operate alongside substantial local autonomy. In each of these examples, the provinces/states/regions retain authority over implementation while participating in shared national or collective price negotiation frameworks.
The U.S. already has partial examples of joint or centralized negotiation, including Medicare drug price negotiation and Medicaid’s supplemental rebate system, as well as newer multipayer initiatives such as the Center for Medicare and Medicaid Innovation’s state-based demonstration for sickle cell disease.
For the U.S., federal leadership, multistate collaboratives, or alliances of public and private payers could further consolidate bargaining power, reduce administrative burden, and support more predictable pricing. Several interviewees stressed that coordination is as much about building trust among payers with competing interests as about formal structures — a reminder that U.S. reforms would require not only legal mechanisms but also willingness among payers to share bargaining leverage.
Risk-sharing and outcome-linked models can balance early access and fiscal accountability.
Several countries have created pathways for early access to high-cost medicines while retaining mechanisms for reassessment. In Germany, early access is embedded directly within the main reimbursement pathway. Under the AMNOG framework, new medicines enter the market immediately at manufacturer-set prices, followed by a mandatory benefit assessment and centralized price negotiation. This ensures rapid access while preserving a formal mechanism for reassessment and price correction. However, it has been criticized for allowing high initial prices and relying heavily on early clinical evidence, prompting calls for stronger postmarket data requirements, particularly for advanced therapies.
England offers a contrasting example. When standard HTA processes cannot readily accommodate uncertainty, dedicated mechanisms fill the gap. The Cancer Drugs Fund — restructured in 2016 after critiques of the earlier version that operated outside NICE — and the newer Innovative Medicines Fund are mechanisms for therapies that cannot be readily approved through standard HTA processes. These schemes provide time-limited access while generating real-world evidence to inform future coverage and pricing decisions, effectively institutionalizing temporary reimbursement as a policy tool.
In parallel, risk-sharing and Managed Entry Agreements (MEAs) have evolved from pilots into standard tools in many countries. These agreements — which include confidential financial arrangements such as rebates, price–volume agreements, and expenditure caps — are widely used to manage budget impact for high-cost therapies. In Sweden, collective regional negotiations expand access to orphan drugs for rare diseases, but these remain primarily financial rather than outcome-linked. France and England similarly rely predominantly on financial agreements, with only limited use of true performance-based contracts.
Despite sustained policy interest, outcome-linked agreements remain rare due to data limitations (discussed further below), administrative burden, and difficulties in attributing real-world outcomes. Several interviewees noted that even non-outcome-based MEAs require significant administrative effort, raising questions about scalability. Performance-based models were often described as aspirational unless health systems develop more robust data systems for tracking patient-level outcomes.
An emerging but underdeveloped dimension of these models is “managed exit” — mechanisms for withdrawing coverage or renegotiating prices if therapies fail to demonstrate real-world value. This aligns with the concept of coverage with evidence generation, in which temporary access is conditional on ongoing evidence development and reassessment. Without credible exit strategies, risk-sharing frameworks may become one-sided.
For the United States, these examples suggest the potential value of more structured pathways for conditional access to novel high-cost therapies. Time-limited reimbursement mechanisms, MEAs, and formal reassessment processes could help balance earlier patient access with greater fiscal accountability and stronger incentives for evidence generation.
Investing in data and evidence infrastructure is essential.
The feasibility of outcome-based reimbursement depends on robust data systems. Countries with national disease registries and experience linking clinical and administrative data, such as Sweden, demonstrate the infrastructure that outcome-linked agreements would require — yet even there, current practice has not moved beyond financial agreements.
Stakeholders noted that data investment was often politically harder to secure than funding for medicines themselves. Several cited the absence of a clear institutional home for data governance, unresolved privacy concerns, and the lack of short-term political returns from infrastructure spending. Germany’s ongoing reforms reflect a growing emphasis on postlaunch evidence generation as a foundation for more dynamic pricing.
Where outcome data collection is limited, financial MEAs do not reveal whether the therapies they cover deliver clinical value. In the U.S., large insurers and integrated data platforms possess extensive claims and utilization data, but fragmentation of data systems across payers and the absence of standardized outcome tracking remain major barriers. Investing in interoperable registries, claims-linkage, and real-world evidence infrastructure, alongside clearer governance frameworks, is essential to support more sophisticated risk-sharing strategies. Without these capabilities, policymakers will remain reliant on blunt cost-control measures rather than more precise, value-informed financing tools.
Equity and patient protection should be explicit policy objectives.
Across all six countries, interviewees emphasized that protecting patients and ensuring equity — access to needed medicines based on clinical need rather than ability to pay — are explicit policy objectives. Rather than allowing individual purchasing power to rule, each nation has embedded these goals in system design, with equity achieved primarily through financial protection and risk pooling.
In practice, patients in these countries generally face low or capped out-of-pocket payments for medicines included on public benefit lists. High-cost medicines are typically financed through national or pooled mechanisms such as special funding pathways, negotiated confidential discounts, or dedicated budgets. Affordability, in other words, is treated as a system-level responsibility. While not all new medicines are covered, and access is often subject to eligibility criteria, patients who meet those criteria are largely shielded from the financial toxicity frequently observed in the U.S.
For the U.S., adapting these approaches would require not only technical reforms to benefit design and pricing but also a commitment to patient affordability as a guiding principle — a structural commitment the U.S. currently lacks. Without such a shift, even sophisticated negotiation or value-based purchasing tools are unlikely to fully address equity gaps in access to high-cost therapies.
Conclusion
Several lessons emerge that provide actionable guidance for U.S. policy adaptation, summarized in Appendix 2. In particular, centralized or coordinated negotiation, along with the selective use of outcome-linked reimbursement, could improve affordability while maintaining incentives for innovation. Investments in data infrastructure and robust real-world evidence systems are essential to enable more sophisticated risk-sharing arrangements, and explicit equity safeguards can help ensure timely access for the patients who need them most.
Adapting these approaches to the U.S. context presents both challenges and opportunities. The fragmentation of the payer landscape, the lack of interoperable systems for collecting and sharing real-world evidence, and resistance to centralized government negotiation are major constraints. Existing legal and regulatory frameworks, including antitrust considerations, may also limit coordination among private payers, reinforcing fragmentation in purchasing power. Clear federal or multipayer frameworks, investment in evidence infrastructure, and enforcement mechanisms are prerequisites for success. Stakeholder alignment among manufacturers, payers, and providers is also critical to reduce the risk of cost shifting or unintended barriers to care.
Across the countries studied, stakeholders consistently emphasized that affordable access to novel high-cost medicines depends on early alignment, transparent communication, and adaptive frameworks. Pilot programs and phased adoption can help the U.S. test models under real-world conditions while mitigating unintended consequences — with explicit affordability protections, critical for uptake and patient trust, as a core design principle.
In conclusion, rising spending on high-cost medicines in the United States threatens both affordability and access. Lessons from peer countries demonstrate that intentional policy design combining negotiation power, evidence-informed decision-making, flexible financing tools, and equity safeguards can support sustainable access to transformative therapies. Careful adaptation, pilot testing, and broad stakeholder engagement will be critical to building a durable, value-driven framework for the United States.

